
 |
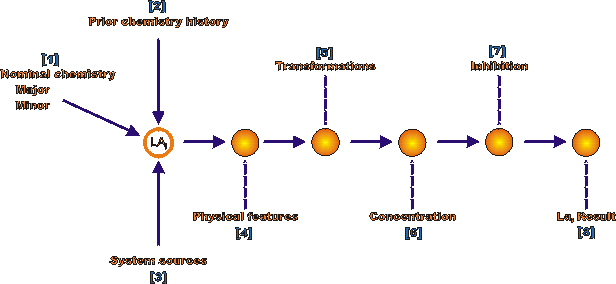
Figure 2 Steps for defining chemical environments applied to a location for analysis, LAi
[1] "Nominal Chemistry" refers to the bulk chemistry. For components exposed to ordinary air atmospheres, the "Major" elements mean humid air. The "Minor" elements refer to industrial contaminants such as SO2 and NO2.
[2] "Prior Chemistry History" refers to exposures to environmental species that might still reside on the surfaces or inside crevices. For example, some subcomponents are cleaned in HF + HNO3 and such environments, unless thoroughly removed, remain on surfaces or within intergranular interstices that are produced by the corrosive action of the cleaning.
[3] "System Sources" refers to those environments that do not come directly from a component but from outside. In concrete, this would be the salt from winter ice removal; whereas, the direct nominal environment is that due to the inherent chemistry of the water leached from concrete. Again, in concrete this would include the phenomenon of "carbonation" that lowers the pH of the concrete and promotes increased corrosion of reinforcing steel.
[4] "Physical Features" includes occluded geometries, flow, and long range electrochemical cells.
[5] "Transformations" refers, for one example, to microbial actions: changing organic materials, such as grease, to organic acids; changing relatively innocuous sulfates to sulfides that accelerate hydrogen entry and increase corrosion rates. Transformations also include the action of long range electrochemical cells that produce acidic and alkaline species at anodic and cathodic sites. The use of hydrazine, which is normally added to minimize he concentration of oxygen, can reduce sulfate to sulfide.
[6] "Concentration" refers to accumulations much greater than that in the bulk environment due to various actions of wetting and drawing, evaporation, potential gradients, and crevices actions that prevent dilution.
[7] "Inhibition" refers to actions taken to minimize corrosive actions. This usually involves additions of oxygen scavengers or species that interfere directly with anodic or cathodic reactions.
Once these are identified and quantified, they provide inputs [8] to the LAM discussed in connection with Figure 4.
Lifetime Prediction, Roger W. Staehle, Adjunct Professor, Department of Chemical Engineering and Materials Science, University of Minnesota, Staehle Consulting Co.