Dental amalgam, in widespread use for over 150 years, is one of
the oldest materials used in oral health care. Its use extends beyond that of
most drugs, and is predated in dentistry only by the use of gold. Dental amalgam
is the end result of mixing approximately equal parts of elemental liquid
mercury (43 to 54 percent) and an alloy powder (57 to 46 percent) composed of
silver, tin, copper, and sometimes smaller amounts of zinc, palladium or indium.
Because of a general decline of dental caries among school children and young
adults, the use of dental amalgam began to decrease in the 1970's.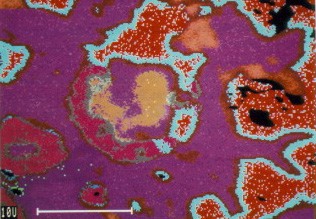
There are also changes in patterns of dental caries, largely the result of topical and systemic fluoride, sealant use, improved oral hygiene practices and products, and possibly dietary modifications. In 1990, over 200 million restorative procedures were provided in the USA. Of these, dental amalgam accounted for roughly 96 million, a 38 percent reduction since 1979. This trend is expected to continue. There are also reports that carious lesions today are generally smaller, easier to treat, and managed by more conservative treatment that retains tooth structure. Because of this decrease in the frequency and size of dental caries, there has been a relative increase in the use of alternative dental restorative materials.
The most commonly used and less expensive of the alternative materials, however, cannot be used for large lesions and need more frequent replacement. Also, there are currently many serviceable dental amalgam restorations that will need replacing in the future. Approximately 70 percent of the restorations placed annually are replacements. Most of these replacements will require amalgam or other metallic materials, because composite materials often lack sufficient strength or durability to be considered adequate substitutes. (reference).
Many blended or dispersed phase high Cu amalgams show excellent clinical performance in long term clinical trials. However, little is known concerning the microstructural changes that occur during clinical use of these systems. The picture on the right shows the in vivo corrosion attack with red and aqua products that penetrate the amalgam withcompositional changes in alloy particles and matrix indicated in shades of purple. (photo courtesy)
|
|
 |